
Contributions
Abstract: S284
Type: Oral Presentation
Session title: Fungal, bacterial and viral infections in hematology
Background
Chimeric antigen receptor T cells (CAR-T) cells targeting CD19, demonstrate highly effective anti-tumor response in Diffuse Large B Cell Lymphoma (DLBCL). However, can result in significant side effects such as prolong neutropenia and infections. The incidence of infections in the setting of real world data hasn’t been completely identified.
Aims
To perform a comprehensive analysis of infection rate and profile in the first month after CAR-T cells in a cohort of infirm patients treated with commercially available CAR-T cells.
Methods
This is a retrospective, single center study conducted in the Bone marrow transplant unit, Tel Aviv, analyzing the infection rate in consecutive patients with DLBCL who were treated with commercially available axicabtagene ciloleucel or tisagenlecleucel. Following a local protocol, all patients were treated with prophylaxis with ciprofloxacin and fluconazole when neutrophil counts decreased below 0.5*103/μl. Acyclovir was administered at conditioning initiation. Microbiology and clinical documented infections (MDI and CDI, respectively) were defined according to the ECIL guidelines. Following white blood cell recovery , patients carried out weekly full blood count, and monthly CMV and HHV-6. A logistic regression was performed for the association between baseline characteristics and documented infections.
Results
From June 2019 to December 2020, we included 60 consecutive patients with DLBCL treated with axicabtagene ciloleucel (n=16, 27%) or tisagenlecleucel (n=44, 73%). The median age was 69.3 (range, 19.8-85.2 ) years and ECOG-Performance status(PS) was 2-3 in most patients (58%). Broad spectrum antibiotics was administered to patients experiencing neutropenic fever (n=53, 88%). Overall infections were noted in 27/60 patients (45%).Bacterial infections were seen in 16 patients (27%) and included CDI in 7 (Pneumonia, n=5; periodontal infection, n=1 and cellulitis, n=1) and MDI in 9 patients (Gram negative rod bacteremia (GNR), n= 5 ; Gram positive cocci(GPC) bacteremia, n= 3; Both GNR and GPC, n=1). Viral infection was described in 14 patients (23%). The most common viral infection was CMV reactivation (n=10, 17%) leading to initiation of anti-CMV treatment in 6 among these patients. None had CMV disease. HHV-6 was positive in 3 patients (5%) with one of these patients presenting with acute encephalitis. Other viral infections reported were RSV (n=2)COVID-19 (n=1). No fungal infection was documented. Incidence of documented infections was higher in patients with CRS/ICANS compared with patients without [ 23/44 (52%) vs 4/16 (25%), p=0.005 ]. While CRP levels predicted documented infections (p= 0.041), ferritin blood levels did not (p=0.130).
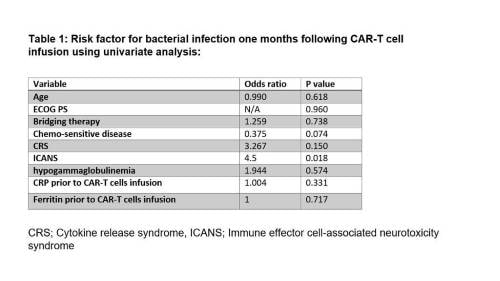
In univariate analysis, ICANS (OR= 4.5, p = 0.018) was associated with higher incidence of bacterial infection while there was a trend for lower incidence of bacterial infections in patients with chemo-sensitive disease to bridging therapy (OR= 0.375, p= 0.074)(Table 1). Age or ECOG-PS were not associated with increased risk of bacterial infection. Patients with documented infection had an increase in hospitalization days compared to those without documented infection (26.44 vs 21.7 days, p= 0.085).
Conclusion
Infections were common in the first month following CAR-T cell administration, however were not increased in elderly patients or those presenting with poorer PS. Patients refractory to bridging therapy and ICANS should be monitored cautiously for the occurrence of infections. CMV monitoring should also be considered.
Keyword(s): CAR-T, Infection, Lymphoma
Abstract: S284
Type: Oral Presentation
Session title: Fungal, bacterial and viral infections in hematology
Background
Chimeric antigen receptor T cells (CAR-T) cells targeting CD19, demonstrate highly effective anti-tumor response in Diffuse Large B Cell Lymphoma (DLBCL). However, can result in significant side effects such as prolong neutropenia and infections. The incidence of infections in the setting of real world data hasn’t been completely identified.
Aims
To perform a comprehensive analysis of infection rate and profile in the first month after CAR-T cells in a cohort of infirm patients treated with commercially available CAR-T cells.
Methods
This is a retrospective, single center study conducted in the Bone marrow transplant unit, Tel Aviv, analyzing the infection rate in consecutive patients with DLBCL who were treated with commercially available axicabtagene ciloleucel or tisagenlecleucel. Following a local protocol, all patients were treated with prophylaxis with ciprofloxacin and fluconazole when neutrophil counts decreased below 0.5*103/μl. Acyclovir was administered at conditioning initiation. Microbiology and clinical documented infections (MDI and CDI, respectively) were defined according to the ECIL guidelines. Following white blood cell recovery , patients carried out weekly full blood count, and monthly CMV and HHV-6. A logistic regression was performed for the association between baseline characteristics and documented infections.
Results
From June 2019 to December 2020, we included 60 consecutive patients with DLBCL treated with axicabtagene ciloleucel (n=16, 27%) or tisagenlecleucel (n=44, 73%). The median age was 69.3 (range, 19.8-85.2 ) years and ECOG-Performance status(PS) was 2-3 in most patients (58%). Broad spectrum antibiotics was administered to patients experiencing neutropenic fever (n=53, 88%). Overall infections were noted in 27/60 patients (45%).Bacterial infections were seen in 16 patients (27%) and included CDI in 7 (Pneumonia, n=5; periodontal infection, n=1 and cellulitis, n=1) and MDI in 9 patients (Gram negative rod bacteremia (GNR), n= 5 ; Gram positive cocci(GPC) bacteremia, n= 3; Both GNR and GPC, n=1). Viral infection was described in 14 patients (23%). The most common viral infection was CMV reactivation (n=10, 17%) leading to initiation of anti-CMV treatment in 6 among these patients. None had CMV disease. HHV-6 was positive in 3 patients (5%) with one of these patients presenting with acute encephalitis. Other viral infections reported were RSV (n=2)COVID-19 (n=1). No fungal infection was documented. Incidence of documented infections was higher in patients with CRS/ICANS compared with patients without [ 23/44 (52%) vs 4/16 (25%), p=0.005 ]. While CRP levels predicted documented infections (p= 0.041), ferritin blood levels did not (p=0.130).
In univariate analysis, ICANS (OR= 4.5, p = 0.018) was associated with higher incidence of bacterial infection while there was a trend for lower incidence of bacterial infections in patients with chemo-sensitive disease to bridging therapy (OR= 0.375, p= 0.074)(Table 1). Age or ECOG-PS were not associated with increased risk of bacterial infection. Patients with documented infection had an increase in hospitalization days compared to those without documented infection (26.44 vs 21.7 days, p= 0.085).

Conclusion
Infections were common in the first month following CAR-T cell administration, however were not increased in elderly patients or those presenting with poorer PS. Patients refractory to bridging therapy and ICANS should be monitored cautiously for the occurrence of infections. CMV monitoring should also be considered.
Keyword(s): CAR-T, Infection, Lymphoma


