
Contributions
Abstract: PB2255
Type: Publication Only
Background
Sclerostin, a product of SOST gene, is a main negative regulator of bone metabolism which exerts its properties through inhibition of WNT signaling pathway in osteoblasts. WNT signaling pathway is implicated in pathogenesis of Philadelphia chromosome negative myeloproliferative neoplasms (Ph- MPNs). These diseases are characterized by remodelling of bone marrow (BM) stroma and development of BM fibrosis/osteosclerosis during course of the disease.
Aims
We aimed to investigate Sclerostin/SOST expression in BM tissue of patients with primary (PMF) and secondary myelofibrosis (SMF) and to assess its clinical correlations.
Methods
Using immunohistochemistry (IHC) and real-time-polymerase-chain-reaction (RT-PCR) we investigated Sclerostin/SOST expression in totally 66 diseased (51 PMF, 15 SMF) and 18 control BM samples. Sclerostin expression measured by IHC was expressed as a percentage of positive cells. SOST expression measured by RT-PCR was normalized to Abl and expressed as a ΔCT value. Samples were collected retrospectively in period from 2006 to 2016. Correlations with clinical parameters were made. The Mann Whitney U test, the Spearman rank correlation, the log-rank test and the Cox regression analysis were used. The ROC curve analysis was used to define optimal cut-off values for survival. P values <0.05 were considered to be statistically significant.
Results
Median age of patients was 67 years, 59.2% were males. Median follow up of our cohort was 74 months. Sclerostin/SOST expression did not significantly differ between healthy and diseased patients, nor between PMF and SMF. However, higher Sclerostin expression in myelofibrosis patients was significantly correlated with lower percentage of circulatory blasts (Rho -0.28, P=0.042) and transfusion dependency (P=0.049). Higher SOST expression in diseased patients was similarly significantly correlated to lower percentage of circulatory blasts (Rho -0.44, P=0.042), but also higher platelets (Rho 0.4, P=0.031) and smaller spleen size (Rho -0.6, P=0.001). We found no significant association of Sclerostin/SOST expression with driver mutations or degree of bone marrow fibrosis.
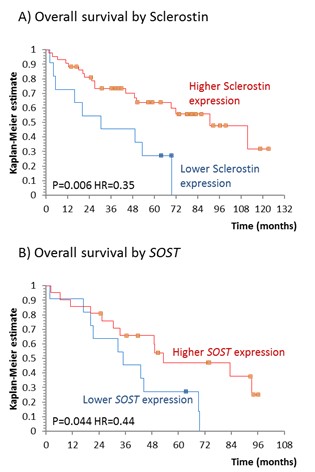
Patients with higher Sclerostin expression measured by IHC (HR=0.35, P=0.006) and SOST expression measured by RT-PCR (HR=0.44, P=0.044) had better overall survival than patients presenting with lower Sclerostin/SOST expression as shown in a Figure. This association remained significant for SOST (HR=0.21, P=0.025) after adjusting for age, gender and circulatory blasts (HR=1.06, P=0.002).
Conclusion
Sclerostin expression might affect stem cell mobilization. Patients with higher Sclerostin/SOST expression experienced improved survival, effect which might be prognostically independent of reduction in circulatory blasts and which is probably mediated through canonical WNT inhibition. Our findings emphasize the role of bone metabolism regulating cytokines, such as Sclerostin, in pathogenesis of Ph- MPNs and suggest that WNT inhibition might be an interesting therapeutic approach in myelofibrosis patients.
Session topic: 15. Myeloproliferative neoplasms – Biology & Translational Research
Keyword(s): prognosis, Bone microenvironment, Cytokine, Myelofibrosis
Abstract: PB2255
Type: Publication Only
Background
Sclerostin, a product of SOST gene, is a main negative regulator of bone metabolism which exerts its properties through inhibition of WNT signaling pathway in osteoblasts. WNT signaling pathway is implicated in pathogenesis of Philadelphia chromosome negative myeloproliferative neoplasms (Ph- MPNs). These diseases are characterized by remodelling of bone marrow (BM) stroma and development of BM fibrosis/osteosclerosis during course of the disease.
Aims
We aimed to investigate Sclerostin/SOST expression in BM tissue of patients with primary (PMF) and secondary myelofibrosis (SMF) and to assess its clinical correlations.
Methods
Using immunohistochemistry (IHC) and real-time-polymerase-chain-reaction (RT-PCR) we investigated Sclerostin/SOST expression in totally 66 diseased (51 PMF, 15 SMF) and 18 control BM samples. Sclerostin expression measured by IHC was expressed as a percentage of positive cells. SOST expression measured by RT-PCR was normalized to Abl and expressed as a ΔCT value. Samples were collected retrospectively in period from 2006 to 2016. Correlations with clinical parameters were made. The Mann Whitney U test, the Spearman rank correlation, the log-rank test and the Cox regression analysis were used. The ROC curve analysis was used to define optimal cut-off values for survival. P values <0.05 were considered to be statistically significant.
Results
Median age of patients was 67 years, 59.2% were males. Median follow up of our cohort was 74 months. Sclerostin/SOST expression did not significantly differ between healthy and diseased patients, nor between PMF and SMF. However, higher Sclerostin expression in myelofibrosis patients was significantly correlated with lower percentage of circulatory blasts (Rho -0.28, P=0.042) and transfusion dependency (P=0.049). Higher SOST expression in diseased patients was similarly significantly correlated to lower percentage of circulatory blasts (Rho -0.44, P=0.042), but also higher platelets (Rho 0.4, P=0.031) and smaller spleen size (Rho -0.6, P=0.001). We found no significant association of Sclerostin/SOST expression with driver mutations or degree of bone marrow fibrosis.
Patients with higher Sclerostin expression measured by IHC (HR=0.35, P=0.006) and SOST expression measured by RT-PCR (HR=0.44, P=0.044) had better overall survival than patients presenting with lower Sclerostin/SOST expression as shown in a Figure. This association remained significant for SOST (HR=0.21, P=0.025) after adjusting for age, gender and circulatory blasts (HR=1.06, P=0.002).

Conclusion
Sclerostin expression might affect stem cell mobilization. Patients with higher Sclerostin/SOST expression experienced improved survival, effect which might be prognostically independent of reduction in circulatory blasts and which is probably mediated through canonical WNT inhibition. Our findings emphasize the role of bone metabolism regulating cytokines, such as Sclerostin, in pathogenesis of Ph- MPNs and suggest that WNT inhibition might be an interesting therapeutic approach in myelofibrosis patients.
Session topic: 15. Myeloproliferative neoplasms – Biology & Translational Research
Keyword(s): prognosis, Bone microenvironment, Cytokine, Myelofibrosis


