
Contributions
Abstract: S425
Type: Oral Presentation
Presentation during EHA22: On Saturday, June 24, 2017 from 12:15 - 12:30
Location: Hall D
Background
Bosutinib (BOS) is a potent, dual SRC/ABL tyrosine kinase inhibitor approved for treatment of adults with Philadelphia chromosome–positive (Ph+) chronic myeloid leukemia (CML) resistant or intolerant to prior therapy.
Aims
To assess the efficacy and safety of BOS versus imatinib (IM) for first-line treatment of chronic phase (CP) CML in the BFORE trial (NCT02130557).
Methods
In this ongoing, multinational, phase 3, open-label study, 536 patients with newly diagnosed CP CML were randomized 1:1 to BOS 400 mg once daily (n=268) or IM 400 mg once daily (n=268 [3 not treated]). Informed consent was obtained from all patients. Per protocol, efficacy was assessed in a modified intent-to-treat (mITT) population of 487 Ph+ patients (BOS, n=246; IM, n=241) with e13a2/e14a2 transcripts; Ph– patients and those with unknown Ph status and/or BCR-ABL transcript type were excluded from this population.
Results
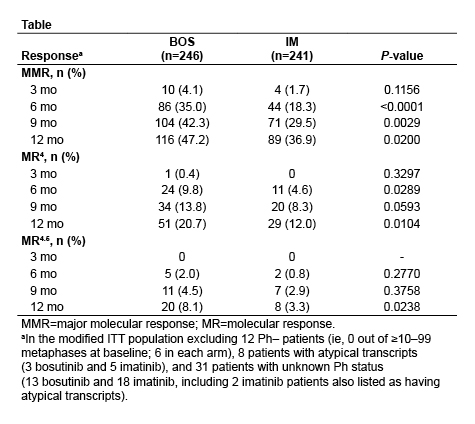
After ≥12 months of follow-up, 78.0% of BOS and 73.2% of IM patients remain on treatment with median treatment durations of 14.1 months and 13.8 months, respectively. Major molecular response (MMR) rate at 12 months (primary endpoint) was significantly higher with BOS versus IM in the mITT population (47.2% vs 36.9%; P=0.02) as well as in the ITT population of all randomized patients (46.6% vs 36.2%; P<0.02). In the mITT population, time to MMR was shorter for BOS (hazard ratio=1.34 based on cumulative incidence; P<0.02). Rate of complete cytogenetic response (CCyR) by 12 months was also significantly higher with BOS versus IM (77.2% vs 66.4%; P<0.008), with time to CCyR shorter for BOS (hazard ratio=1.38; P≤0.001). Rate of BCR-ABL transcripts ≤10% (Intl. Scale) at 3 months was higher with BOS versus IM (75.2% vs 57.3%; P<0.001); rates of deep molecular response over time were also generally higher with BOS (Table). Results for molecular endpoints were similar in the ITT population. The only baseline characteristic identified as a significant predictor of MMR at 12 months besides treatment arm was Sokal risk group (high vs low; P<0.0001 and intermediate vs low; P<0.05 [mITT]). On-treatment progression to accelerated or blast phase occurred in 4 patients (1.6%) receiving BOS and 6 patients (2.5%) receiving IM in the mITT population. One BOS-treated and 4 IM-treated patients discontinued treatment due to progression to accelerated or blast phase. Among all treated patients, there were no deaths within 28 days of last dose of BOS and 4 with IM. Safety data for treated patients were consistent with the known safety profiles of BOS and IM. Discontinuation due to drug-related toxicity occurred with 12.7% of BOS patients and 8.7% of IM patients. Grade ≥3 diarrhea (7.8% vs 0.8%) and increased alanine (19.0% vs 1.5%) and aspartate (9.7% vs 1.9%) aminotransferase levels were more common with BOS. Cardiovascular, peripheral vascular, and cerebrovascular events were infrequent in both groups (all grades: 3.0%, 1.5%, and 0% BOS vs 0.4%, 1.1%, and 0.4% IM; grade ≥3: 1.5%, 0%, and 0% BOS vs 0%, 0%, and 0.4% IM).
Conclusion
Patients on BOS had significantly higher rates of 12-month MMR and CCyR and achieved responses faster than those on IM. Consistent with the known safety profile, higher incidences of gastrointestinal events and transaminase elevations were observed with BOS. Primary results from this study suggest BOS may be an important treatment option for patients with newly diagnosed CP CML.
Session topic: 8. Chronic myeloid leukemia - Clinical
Keyword(s): Tyrosine kinase inhibitor, Randomized, Clinical Trial, Chronic myeloid leukemia
Abstract: S425
Type: Oral Presentation
Presentation during EHA22: On Saturday, June 24, 2017 from 12:15 - 12:30
Location: Hall D
Background
Bosutinib (BOS) is a potent, dual SRC/ABL tyrosine kinase inhibitor approved for treatment of adults with Philadelphia chromosome–positive (Ph+) chronic myeloid leukemia (CML) resistant or intolerant to prior therapy.
Aims
To assess the efficacy and safety of BOS versus imatinib (IM) for first-line treatment of chronic phase (CP) CML in the BFORE trial (NCT02130557).
Methods
In this ongoing, multinational, phase 3, open-label study, 536 patients with newly diagnosed CP CML were randomized 1:1 to BOS 400 mg once daily (n=268) or IM 400 mg once daily (n=268 [3 not treated]). Informed consent was obtained from all patients. Per protocol, efficacy was assessed in a modified intent-to-treat (mITT) population of 487 Ph+ patients (BOS, n=246; IM, n=241) with e13a2/e14a2 transcripts; Ph– patients and those with unknown Ph status and/or BCR-ABL transcript type were excluded from this population.
Results
After ≥12 months of follow-up, 78.0% of BOS and 73.2% of IM patients remain on treatment with median treatment durations of 14.1 months and 13.8 months, respectively. Major molecular response (MMR) rate at 12 months (primary endpoint) was significantly higher with BOS versus IM in the mITT population (47.2% vs 36.9%; P=0.02) as well as in the ITT population of all randomized patients (46.6% vs 36.2%; P<0.02). In the mITT population, time to MMR was shorter for BOS (hazard ratio=1.34 based on cumulative incidence; P<0.02). Rate of complete cytogenetic response (CCyR) by 12 months was also significantly higher with BOS versus IM (77.2% vs 66.4%; P<0.008), with time to CCyR shorter for BOS (hazard ratio=1.38; P≤0.001). Rate of BCR-ABL transcripts ≤10% (Intl. Scale) at 3 months was higher with BOS versus IM (75.2% vs 57.3%; P<0.001); rates of deep molecular response over time were also generally higher with BOS (Table). Results for molecular endpoints were similar in the ITT population. The only baseline characteristic identified as a significant predictor of MMR at 12 months besides treatment arm was Sokal risk group (high vs low; P<0.0001 and intermediate vs low; P<0.05 [mITT]). On-treatment progression to accelerated or blast phase occurred in 4 patients (1.6%) receiving BOS and 6 patients (2.5%) receiving IM in the mITT population. One BOS-treated and 4 IM-treated patients discontinued treatment due to progression to accelerated or blast phase. Among all treated patients, there were no deaths within 28 days of last dose of BOS and 4 with IM. Safety data for treated patients were consistent with the known safety profiles of BOS and IM. Discontinuation due to drug-related toxicity occurred with 12.7% of BOS patients and 8.7% of IM patients. Grade ≥3 diarrhea (7.8% vs 0.8%) and increased alanine (19.0% vs 1.5%) and aspartate (9.7% vs 1.9%) aminotransferase levels were more common with BOS. Cardiovascular, peripheral vascular, and cerebrovascular events were infrequent in both groups (all grades: 3.0%, 1.5%, and 0% BOS vs 0.4%, 1.1%, and 0.4% IM; grade ≥3: 1.5%, 0%, and 0% BOS vs 0%, 0%, and 0.4% IM).

Conclusion
Patients on BOS had significantly higher rates of 12-month MMR and CCyR and achieved responses faster than those on IM. Consistent with the known safety profile, higher incidences of gastrointestinal events and transaminase elevations were observed with BOS. Primary results from this study suggest BOS may be an important treatment option for patients with newly diagnosed CP CML.
Session topic: 8. Chronic myeloid leukemia - Clinical
Keyword(s): Tyrosine kinase inhibitor, Randomized, Clinical Trial, Chronic myeloid leukemia


