YOU DON'T KNOW JAK: A PROGRAMMED RIBOSOMAL FRAMESHIFTING DEFECT POTENTIATES THE TRANSFORMING ACTIVITY OF THE JAK2-V617F MUTATION
(Abstract release date: 05/18/17)
EHA Library. SULIMA S. 06/24/17; 181704; S417

Sergey SULIMA
Contributions
Contributions
Abstract
Abstract: S417
Type: Oral Presentation
Presentation during EHA22: On Saturday, June 24, 2017 from 11:30 - 11:45
Location: Hall C
Background
The JAK-STAT pathway is a critical controller of cellular proliferation, differentiation, survival and apoptosis in response to external stimuli. Promiscuous activation of this pathway is an important driver in the pathogenesis of BCR/ABL-negative chronic myeloproliferative neoplasms. The JAK2-V617F allele is the most common and characterized mutation linked to this class of leukemia. The increased activation of JAK-STAT signaling in JAK2-V617F cells can be partially explained by increased JAK2 autophosphorylation. It is unclear however if these effects are sufficient to fully account for the strong activation of the JAK-STAT pathway induced by JAK2-V617F.
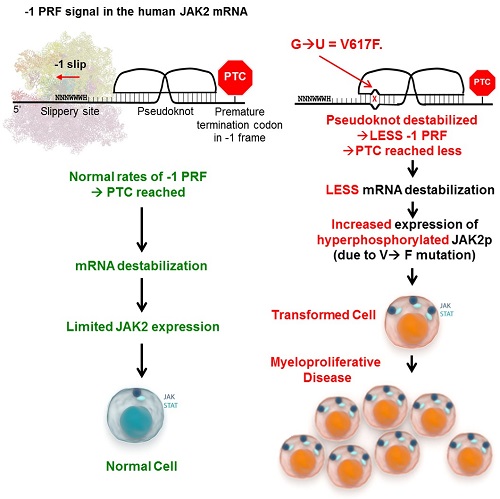
We recently described programmed -1 ribosomal frameshifting (-1 PRF) as a novel mechanism regulating the expression of ~10% of human genes, including cytokine receptors (Belew AT et al, Nature, 2014). In this process, cis-acting mRNA elements (-1 PRF signals, which consist of a slippery site followed by a pseudoknot) direct translating ribosomes to slip by one base in the 5’ direction, establishing a new reading frame. This directs ribosomes towards premature termination codons, resulting in destabilization of the -1 PRF signal-containing mRNA via nonsense-mediated mRNA decay (Figure). There is thus an inverse relationship between -1 PRF efficiency and mRNA stability.
Aims
To investigate whether the JAK2-V617F mutation, shown here to be located in the pseudoknot of a -1 PRF signal in the JAK2 mRNA, impacts disease progression through ablation of -1 PRF.
Methods
Computationally predicted -1 PRF signals were validated using dual luciferase reporters and proteomic analysis of a -1 PRF fusion protein. -1 PRF as well as mRNA abundance and decay were assayed in HEK293T and HeLa cells. Transformation assays were performed in hEpoR expressing Ba/F3 cells. In vivo experiments were performed in BALB/c mice.
Results
We demonstrate in human cell lines that the JAK2-V617F mutation structurally disrupts the -1 PRF signal in the JAK2 mRNA, leading to ~2-fold lower rates of -1 PRF and increased abundance of the JAK2 mRNA and protein. The transforming potential of a series of mutants designed to manipulate -1 PRF independent of V617F was assayed in a Ba/F3 cell model. Silent protein coding changes in the pseudoknot of the -1 PRF signal at position V617 (V617m) or the slippery site (SSm), both of which reduced frameshifting, increased JAK2 expression and led to transforming activity, albeit less than V617F. Importantly, the V617F+SSm combination conferred an additive effect on cellular transformation. Ba/F3 cells expressing these JAK2 variants were also introduced into mice. Whereas mice injected with wild type JAK2 remained healthy, both V617m and SSm induced similar leukemia phenotypes as V617F and V617F+SSm, with a ~2-fold longer disease latency of 8-10 weeks. Increased JAK2 mRNA abundance in JAK2-V617F homozygous patients as well as the presence of three additional -1 PRF signals in the JAK2 mRNA further suggest a prominent role for -1 PRF in controlling JAK2 production.
Conclusion
We demonstrate that the JAK2-V617F mutation diminishes -1 PRF on the JAK2 transcript, stabilizing the mRNA and increasing JAK2 expression, contributing to its transforming activity in vitro and disease onset in vivo. We suggest that -1 PRF normally provides a layer of control by limiting JAK2 translation. Defective -1 PRF synergizes with the transforming activity of the JAK2-V617F protein by causing its overexpression, explaining why this particular mutation causes such aggressive malignancies. In support of this, the combination of ruxolitinib and an HSP-90 inhibitor, which reduce kinase activity and JAK2 expression respectively, leads to increased therapeutic efficacy in myeloproliferative neoplasms (Bhagwat N et al, Blood, 2014).
Session topic: 15. Myeloproliferative neoplasms - Biology
Abstract: S417
Type: Oral Presentation
Presentation during EHA22: On Saturday, June 24, 2017 from 11:30 - 11:45
Location: Hall C
Background
The JAK-STAT pathway is a critical controller of cellular proliferation, differentiation, survival and apoptosis in response to external stimuli. Promiscuous activation of this pathway is an important driver in the pathogenesis of BCR/ABL-negative chronic myeloproliferative neoplasms. The JAK2-V617F allele is the most common and characterized mutation linked to this class of leukemia. The increased activation of JAK-STAT signaling in JAK2-V617F cells can be partially explained by increased JAK2 autophosphorylation. It is unclear however if these effects are sufficient to fully account for the strong activation of the JAK-STAT pathway induced by JAK2-V617F.
We recently described programmed -1 ribosomal frameshifting (-1 PRF) as a novel mechanism regulating the expression of ~10% of human genes, including cytokine receptors (Belew AT et al, Nature, 2014). In this process, cis-acting mRNA elements (-1 PRF signals, which consist of a slippery site followed by a pseudoknot) direct translating ribosomes to slip by one base in the 5’ direction, establishing a new reading frame. This directs ribosomes towards premature termination codons, resulting in destabilization of the -1 PRF signal-containing mRNA via nonsense-mediated mRNA decay (Figure). There is thus an inverse relationship between -1 PRF efficiency and mRNA stability.
Aims
To investigate whether the JAK2-V617F mutation, shown here to be located in the pseudoknot of a -1 PRF signal in the JAK2 mRNA, impacts disease progression through ablation of -1 PRF.
Methods
Computationally predicted -1 PRF signals were validated using dual luciferase reporters and proteomic analysis of a -1 PRF fusion protein. -1 PRF as well as mRNA abundance and decay were assayed in HEK293T and HeLa cells. Transformation assays were performed in hEpoR expressing Ba/F3 cells. In vivo experiments were performed in BALB/c mice.
Results
We demonstrate in human cell lines that the JAK2-V617F mutation structurally disrupts the -1 PRF signal in the JAK2 mRNA, leading to ~2-fold lower rates of -1 PRF and increased abundance of the JAK2 mRNA and protein. The transforming potential of a series of mutants designed to manipulate -1 PRF independent of V617F was assayed in a Ba/F3 cell model. Silent protein coding changes in the pseudoknot of the -1 PRF signal at position V617 (V617m) or the slippery site (SSm), both of which reduced frameshifting, increased JAK2 expression and led to transforming activity, albeit less than V617F. Importantly, the V617F+SSm combination conferred an additive effect on cellular transformation. Ba/F3 cells expressing these JAK2 variants were also introduced into mice. Whereas mice injected with wild type JAK2 remained healthy, both V617m and SSm induced similar leukemia phenotypes as V617F and V617F+SSm, with a ~2-fold longer disease latency of 8-10 weeks. Increased JAK2 mRNA abundance in JAK2-V617F homozygous patients as well as the presence of three additional -1 PRF signals in the JAK2 mRNA further suggest a prominent role for -1 PRF in controlling JAK2 production.

Conclusion
We demonstrate that the JAK2-V617F mutation diminishes -1 PRF on the JAK2 transcript, stabilizing the mRNA and increasing JAK2 expression, contributing to its transforming activity in vitro and disease onset in vivo. We suggest that -1 PRF normally provides a layer of control by limiting JAK2 translation. Defective -1 PRF synergizes with the transforming activity of the JAK2-V617F protein by causing its overexpression, explaining why this particular mutation causes such aggressive malignancies. In support of this, the combination of ruxolitinib and an HSP-90 inhibitor, which reduce kinase activity and JAK2 expression respectively, leads to increased therapeutic efficacy in myeloproliferative neoplasms (Bhagwat N et al, Blood, 2014).
Session topic: 15. Myeloproliferative neoplasms - Biology
{{ help_message }}
{{filter}}


