
Contributions
Abstract: S413
Type: Oral Presentation
Presentation during EHA22: On Saturday, June 24, 2017 from 11:45 - 12:00
Location: Hall B
Background
Aims
To confirm in a prospective setting the favorable prognosis of advanced stage PET2 negative patients treated with ABVD, as well as the safety and efficacy of escalated BEACOPP given to PET2 positive patients.
Methods
Results
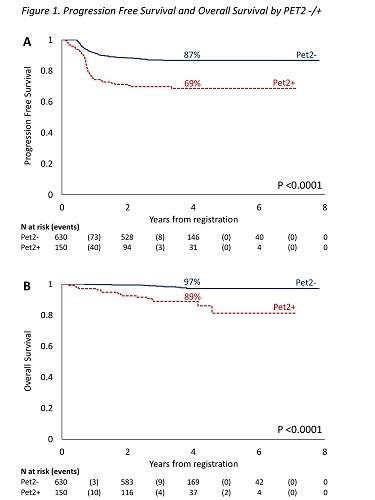
Conclusion
Session topic: 17. Hodgkin lymphoma - Clinical
Keyword(s): PET, Hodgkin's disease
Abstract: S413
Type: Oral Presentation
Presentation during EHA22: On Saturday, June 24, 2017 from 11:45 - 12:00
Location: Hall B
Background
Aims
To confirm in a prospective setting the favorable prognosis of advanced stage PET2 negative patients treated with ABVD, as well as the safety and efficacy of escalated BEACOPP given to PET2 positive patients.
Methods
Results

Conclusion
Session topic: 17. Hodgkin lymphoma - Clinical
Keyword(s): PET, Hodgkin's disease


