ALXN1210, A LONG-ACTING C5 INHIBITOR, RESULTS IN RAPID AND SUSTAINED REDUCTION OF LDH WITH A MONTHLY DOSING INTERVAL IN PATIENTS WITH PNH: PRELIMINARY DATA FROM A DOSE-ESCALATION STUDY
(Abstract release date: 05/19/16)
EHA Library. Lee J. 06/09/16; 135358; LB2247
Disclosure(s): Received honoraria and research funding from and is a consultant for Alexion Pharmaceuticals, Inc.

Prof. Dr. Jong Wook Lee
Contributions
Contributions
Abstract
Abstract: LB2247
Type: Eposter Presentation
Background
ALXN1210, a rationally designed humanized monoclonal antibody to C5, was engineered for an extended duration of complement inhibition. A previous study in healthy volunteers showed immediate, complete, and sustained inhibition of C5. The half-life (t½) of ALXN1210 was >30 days—substantially longer than eculizumab—which facilitates an extended dosing interval of ≥1 mo.
Aims
ALXN1210-PNH-103 is a Phase 1b, multicenter, open-label, intrapatient dose-escalation study (NCT02598583), evaluating the safety, tolerability, and efficacy of 2 IV maintenance dosing regimens of ALXN1210 in patients (pts) with paroxysmal nocturnal hemoglobinuria (PNH) never treated with a complement inhibitor.
Methods
In this interim analysis, 2 cohorts of pts ≥18 y were investigated: pts in Cohort 1 (C1) received either 400- or 600-mg induction doses followed by a 900-mg maintenance dose q4w; pts in Cohort 2 (C2) received 600- and 900-mg induction doses, followed by an 1800-mg maintenance dose q4w. The primary objective was to assess safety and tolerability of ALXN1210. The primary efficacy outcome was change in lactate dehydrogenase (LDH) level. Other endpoints included change in blood transfusions and in hematologic parameters related to PNH.
Results
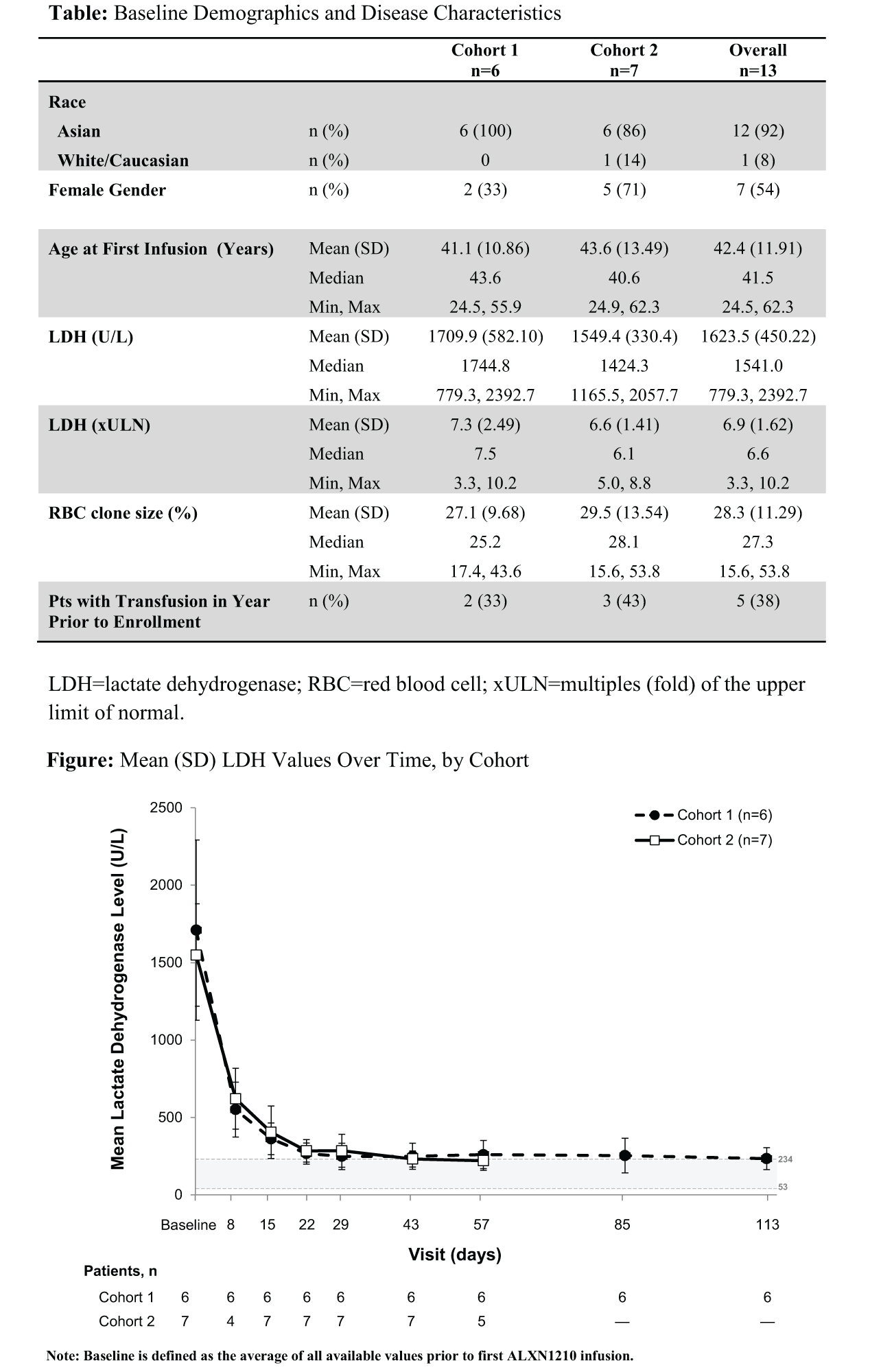
A total of 13 pts consented and enrolled (Table). Median duration of exposure was 3.7 mo for C1 (n=6) and 1.8 mo for C2 (n=7). All pts showed rapid reductions in LDH levels, which were observed at the first evaluable time point (day 8). Decreases in LDH were sustained over 4-mo dose intervals, as shown by mean reductions from baseline in LDH of 85.6% in C1 on day 113 and 83.8% in C2 on day 57, the latest evaluable time points (Figure). Among 5 pts with ≥1 transfusion in the year before treatment (2 in C1, 3 in C2), only 1 patient (in C1, who had received 12 units of packed red blood cells in the prior 6 mo) received a transfusion (2 units) while on ALXN1210. Multiple doses of ALXN1210 resulted in no serious adverse events (SAEs), no infusion site reactions, and no drug discontinuations or AEs leading to withdrawals. The most common treatment-emergent AE (TEAE) was headache (2 pts). Investigators judged 69% of TEAEs to be unrelated to treatment. All related AEs resolved with ongoing ALXN1210 treatment.
Conclusion
ALXN1210-PNH-103 is the first study to demonstrate safety and efficacy of ALXN1210 in pts with PNH. ALXN1210 treatment resulted in rapid reductions in LDH levels in 100% of pts, which were sustained through 2-4 monthly dosing intervals, consistent with the extended t½ of ALXN1210. There was a notable decrease in the need for blood transfusions. These preliminary LDH data suggest that rapid, complete, and sustained complement inhibition with ALXN1210 results in highly effective blockade of intravascular hemolysis. A Phase 2 study examining monthly and longer dosing intervals of ALXN1210 is ongoing.
Session topic: E-poster
Keyword(s): Complement, Inhibitor, Paroxysmal nocturnal hemoglobinuria (PNH), PNH
Type: Eposter Presentation
Background
ALXN1210, a rationally designed humanized monoclonal antibody to C5, was engineered for an extended duration of complement inhibition. A previous study in healthy volunteers showed immediate, complete, and sustained inhibition of C5. The half-life (t½) of ALXN1210 was >30 days—substantially longer than eculizumab—which facilitates an extended dosing interval of ≥1 mo.
Aims
ALXN1210-PNH-103 is a Phase 1b, multicenter, open-label, intrapatient dose-escalation study (NCT02598583), evaluating the safety, tolerability, and efficacy of 2 IV maintenance dosing regimens of ALXN1210 in patients (pts) with paroxysmal nocturnal hemoglobinuria (PNH) never treated with a complement inhibitor.
Methods
In this interim analysis, 2 cohorts of pts ≥18 y were investigated: pts in Cohort 1 (C1) received either 400- or 600-mg induction doses followed by a 900-mg maintenance dose q4w; pts in Cohort 2 (C2) received 600- and 900-mg induction doses, followed by an 1800-mg maintenance dose q4w. The primary objective was to assess safety and tolerability of ALXN1210. The primary efficacy outcome was change in lactate dehydrogenase (LDH) level. Other endpoints included change in blood transfusions and in hematologic parameters related to PNH.
Results
A total of 13 pts consented and enrolled (Table). Median duration of exposure was 3.7 mo for C1 (n=6) and 1.8 mo for C2 (n=7). All pts showed rapid reductions in LDH levels, which were observed at the first evaluable time point (day 8). Decreases in LDH were sustained over 4-mo dose intervals, as shown by mean reductions from baseline in LDH of 85.6% in C1 on day 113 and 83.8% in C2 on day 57, the latest evaluable time points (Figure). Among 5 pts with ≥1 transfusion in the year before treatment (2 in C1, 3 in C2), only 1 patient (in C1, who had received 12 units of packed red blood cells in the prior 6 mo) received a transfusion (2 units) while on ALXN1210. Multiple doses of ALXN1210 resulted in no serious adverse events (SAEs), no infusion site reactions, and no drug discontinuations or AEs leading to withdrawals. The most common treatment-emergent AE (TEAE) was headache (2 pts). Investigators judged 69% of TEAEs to be unrelated to treatment. All related AEs resolved with ongoing ALXN1210 treatment.
Conclusion
ALXN1210-PNH-103 is the first study to demonstrate safety and efficacy of ALXN1210 in pts with PNH. ALXN1210 treatment resulted in rapid reductions in LDH levels in 100% of pts, which were sustained through 2-4 monthly dosing intervals, consistent with the extended t½ of ALXN1210. There was a notable decrease in the need for blood transfusions. These preliminary LDH data suggest that rapid, complete, and sustained complement inhibition with ALXN1210 results in highly effective blockade of intravascular hemolysis. A Phase 2 study examining monthly and longer dosing intervals of ALXN1210 is ongoing.

Session topic: E-poster
Keyword(s): Complement, Inhibitor, Paroxysmal nocturnal hemoglobinuria (PNH), PNH
Abstract: LB2247
Type: Eposter Presentation
Background
ALXN1210, a rationally designed humanized monoclonal antibody to C5, was engineered for an extended duration of complement inhibition. A previous study in healthy volunteers showed immediate, complete, and sustained inhibition of C5. The half-life (t½) of ALXN1210 was >30 days—substantially longer than eculizumab—which facilitates an extended dosing interval of ≥1 mo.
Aims
ALXN1210-PNH-103 is a Phase 1b, multicenter, open-label, intrapatient dose-escalation study (NCT02598583), evaluating the safety, tolerability, and efficacy of 2 IV maintenance dosing regimens of ALXN1210 in patients (pts) with paroxysmal nocturnal hemoglobinuria (PNH) never treated with a complement inhibitor.
Methods
In this interim analysis, 2 cohorts of pts ≥18 y were investigated: pts in Cohort 1 (C1) received either 400- or 600-mg induction doses followed by a 900-mg maintenance dose q4w; pts in Cohort 2 (C2) received 600- and 900-mg induction doses, followed by an 1800-mg maintenance dose q4w. The primary objective was to assess safety and tolerability of ALXN1210. The primary efficacy outcome was change in lactate dehydrogenase (LDH) level. Other endpoints included change in blood transfusions and in hematologic parameters related to PNH.
Results
A total of 13 pts consented and enrolled (Table). Median duration of exposure was 3.7 mo for C1 (n=6) and 1.8 mo for C2 (n=7). All pts showed rapid reductions in LDH levels, which were observed at the first evaluable time point (day 8). Decreases in LDH were sustained over 4-mo dose intervals, as shown by mean reductions from baseline in LDH of 85.6% in C1 on day 113 and 83.8% in C2 on day 57, the latest evaluable time points (Figure). Among 5 pts with ≥1 transfusion in the year before treatment (2 in C1, 3 in C2), only 1 patient (in C1, who had received 12 units of packed red blood cells in the prior 6 mo) received a transfusion (2 units) while on ALXN1210. Multiple doses of ALXN1210 resulted in no serious adverse events (SAEs), no infusion site reactions, and no drug discontinuations or AEs leading to withdrawals. The most common treatment-emergent AE (TEAE) was headache (2 pts). Investigators judged 69% of TEAEs to be unrelated to treatment. All related AEs resolved with ongoing ALXN1210 treatment.
Conclusion
ALXN1210-PNH-103 is the first study to demonstrate safety and efficacy of ALXN1210 in pts with PNH. ALXN1210 treatment resulted in rapid reductions in LDH levels in 100% of pts, which were sustained through 2-4 monthly dosing intervals, consistent with the extended t½ of ALXN1210. There was a notable decrease in the need for blood transfusions. These preliminary LDH data suggest that rapid, complete, and sustained complement inhibition with ALXN1210 results in highly effective blockade of intravascular hemolysis. A Phase 2 study examining monthly and longer dosing intervals of ALXN1210 is ongoing.

Session topic: E-poster
Keyword(s): Complement, Inhibitor, Paroxysmal nocturnal hemoglobinuria (PNH), PNH
Type: Eposter Presentation
Background
ALXN1210, a rationally designed humanized monoclonal antibody to C5, was engineered for an extended duration of complement inhibition. A previous study in healthy volunteers showed immediate, complete, and sustained inhibition of C5. The half-life (t½) of ALXN1210 was >30 days—substantially longer than eculizumab—which facilitates an extended dosing interval of ≥1 mo.
Aims
ALXN1210-PNH-103 is a Phase 1b, multicenter, open-label, intrapatient dose-escalation study (NCT02598583), evaluating the safety, tolerability, and efficacy of 2 IV maintenance dosing regimens of ALXN1210 in patients (pts) with paroxysmal nocturnal hemoglobinuria (PNH) never treated with a complement inhibitor.
Methods
In this interim analysis, 2 cohorts of pts ≥18 y were investigated: pts in Cohort 1 (C1) received either 400- or 600-mg induction doses followed by a 900-mg maintenance dose q4w; pts in Cohort 2 (C2) received 600- and 900-mg induction doses, followed by an 1800-mg maintenance dose q4w. The primary objective was to assess safety and tolerability of ALXN1210. The primary efficacy outcome was change in lactate dehydrogenase (LDH) level. Other endpoints included change in blood transfusions and in hematologic parameters related to PNH.
Results
A total of 13 pts consented and enrolled (Table). Median duration of exposure was 3.7 mo for C1 (n=6) and 1.8 mo for C2 (n=7). All pts showed rapid reductions in LDH levels, which were observed at the first evaluable time point (day 8). Decreases in LDH were sustained over 4-mo dose intervals, as shown by mean reductions from baseline in LDH of 85.6% in C1 on day 113 and 83.8% in C2 on day 57, the latest evaluable time points (Figure). Among 5 pts with ≥1 transfusion in the year before treatment (2 in C1, 3 in C2), only 1 patient (in C1, who had received 12 units of packed red blood cells in the prior 6 mo) received a transfusion (2 units) while on ALXN1210. Multiple doses of ALXN1210 resulted in no serious adverse events (SAEs), no infusion site reactions, and no drug discontinuations or AEs leading to withdrawals. The most common treatment-emergent AE (TEAE) was headache (2 pts). Investigators judged 69% of TEAEs to be unrelated to treatment. All related AEs resolved with ongoing ALXN1210 treatment.
Conclusion
ALXN1210-PNH-103 is the first study to demonstrate safety and efficacy of ALXN1210 in pts with PNH. ALXN1210 treatment resulted in rapid reductions in LDH levels in 100% of pts, which were sustained through 2-4 monthly dosing intervals, consistent with the extended t½ of ALXN1210. There was a notable decrease in the need for blood transfusions. These preliminary LDH data suggest that rapid, complete, and sustained complement inhibition with ALXN1210 results in highly effective blockade of intravascular hemolysis. A Phase 2 study examining monthly and longer dosing intervals of ALXN1210 is ongoing.

Session topic: E-poster
Keyword(s): Complement, Inhibitor, Paroxysmal nocturnal hemoglobinuria (PNH), PNH
{{ help_message }}
{{filter}}


