HIGH EXPRESSION OF AF1Q IS AN ADVERSE PROGNOSTIC FACTOR AND A PREDICTION MARKER OF EXTRAMEDULLARY DISEASE IN MULTIPLE MYELOMA
(Abstract release date: 05/19/16)
EHA Library. Hagiwara S. 06/09/16; 132831; E1282
Disclosure(s): Celgene Corporation: Advisory board member

Dr. Shotaro Hagiwara
Contributions
Contributions
Abstract
Abstract: E1282
Type: Eposter Presentation
Background
Multiple myeloma is an intractable hematological malignancy with various clinical manifestations, especially extramedullary disease (EMD). The prognosis of patients with EMD is extremely poor due to limited treatment options and aggressive nature of EMD, however the mechanism of the progression of EMD is not well known.AF1q is an oncogene which expressed in leukemia cells, located in 1q21. The gene is well known as one of the fusion partners of MLL, and as a poor prognostic factor in acute myeloid leukemia and myelodysplastic syndrome . Recently, we reported that high expression of AF1q results in an autonomous Wnt activity that promotes distant metastasis of breast cancer.
Aims
We hypothesized that high expression of AF1q is a poor prognostic factor of multiple myeloma and associated with EMD.To evaluate our hypothesis, we analyzed the expression of AF1q in patients with multiple myeloma and investigated the impact on the prognosis and the progression of EMD.
Methods
Newly diagnosed multiple myeloma patients in National Center for Global Health and Medicine hospital from January 2001 to March 2013 were studied. Patients were treated with vincristine-adriamycin-dexamethason or bortezomib-dexamethason induction therapy followed by autologous stem cell transplantation using high dose melphalan.The expression of AF1q was evaluated using the immunostaining of bone marrow clot samples at the diagnosis of multiple myeloma. The expression of AF1q was graded from “-“ to “+++”. EMD was diagnosed by pathological examination and/or CT/MRI/PET. The clinical response, progression free survival (PFS), and overall survival (OS) were analyzed using Kaplan Meier method with Log-rank test. Correlation between EMD and AF1q expression was tested by Chi-square test.
Results
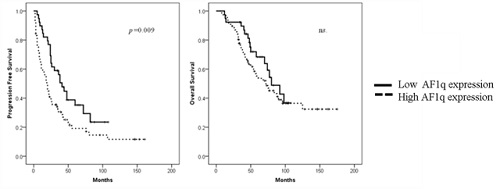
Clinical data and bone marrow clot samples of 117 patients were analyzed. Mean age was 54.9 years old, and 51.9% was male. The grades of AF1q expression were 7 of “-”, 33 of “+”, 35 of “++”, and 42 of “+++”. We defined the cases with “-” and “+” as low expression, “++” and “+++” as high expression. Very good partial response or better was obtained after completion of autologous stem cell transplantation in 42.5% of patients with low AF1q expression and 37.7% of high expression. There was no statistically significant difference. Survival analysis using Log Rank method showed that high expression of AF1q was associated with significantly shorter progression free survival (3 year PFS was 56.2% in low AF1q expression vs. 30.5% in high AF1q expression, p=0.009), but not overall survival (3 year OS was 89.6% in low AF1q expression vs. 73.3% in high AF1q expression, ns. ).EMD was found in 25.0% of patients with low AF1q expression and 44.7% of high expression. Chi-square test showed that the incidence of EMD was significantly higher in patients with high AF1q expression than low expression (p=0.045).
Conclusion
To our knowledge, this is the first study that demonstrates a molecular marker associated with myeloma with EMD. We found that the high expression of AF1q was an adverse prognostic factor in multiple myeloma.
Session topic: E-poster
Keyword(s): Multiple myeloma, Prognostic factor, Stem cell transplant
Type: Eposter Presentation
Background
Multiple myeloma is an intractable hematological malignancy with various clinical manifestations, especially extramedullary disease (EMD). The prognosis of patients with EMD is extremely poor due to limited treatment options and aggressive nature of EMD, however the mechanism of the progression of EMD is not well known.AF1q is an oncogene which expressed in leukemia cells, located in 1q21. The gene is well known as one of the fusion partners of MLL, and as a poor prognostic factor in acute myeloid leukemia and myelodysplastic syndrome . Recently, we reported that high expression of AF1q results in an autonomous Wnt activity that promotes distant metastasis of breast cancer.
Aims
We hypothesized that high expression of AF1q is a poor prognostic factor of multiple myeloma and associated with EMD.To evaluate our hypothesis, we analyzed the expression of AF1q in patients with multiple myeloma and investigated the impact on the prognosis and the progression of EMD.
Methods
Newly diagnosed multiple myeloma patients in National Center for Global Health and Medicine hospital from January 2001 to March 2013 were studied. Patients were treated with vincristine-adriamycin-dexamethason or bortezomib-dexamethason induction therapy followed by autologous stem cell transplantation using high dose melphalan.The expression of AF1q was evaluated using the immunostaining of bone marrow clot samples at the diagnosis of multiple myeloma. The expression of AF1q was graded from “-“ to “+++”. EMD was diagnosed by pathological examination and/or CT/MRI/PET. The clinical response, progression free survival (PFS), and overall survival (OS) were analyzed using Kaplan Meier method with Log-rank test. Correlation between EMD and AF1q expression was tested by Chi-square test.
Results
Clinical data and bone marrow clot samples of 117 patients were analyzed. Mean age was 54.9 years old, and 51.9% was male. The grades of AF1q expression were 7 of “-”, 33 of “+”, 35 of “++”, and 42 of “+++”. We defined the cases with “-” and “+” as low expression, “++” and “+++” as high expression. Very good partial response or better was obtained after completion of autologous stem cell transplantation in 42.5% of patients with low AF1q expression and 37.7% of high expression. There was no statistically significant difference. Survival analysis using Log Rank method showed that high expression of AF1q was associated with significantly shorter progression free survival (3 year PFS was 56.2% in low AF1q expression vs. 30.5% in high AF1q expression, p=0.009), but not overall survival (3 year OS was 89.6% in low AF1q expression vs. 73.3% in high AF1q expression, ns. ).EMD was found in 25.0% of patients with low AF1q expression and 44.7% of high expression. Chi-square test showed that the incidence of EMD was significantly higher in patients with high AF1q expression than low expression (p=0.045).
Conclusion
To our knowledge, this is the first study that demonstrates a molecular marker associated with myeloma with EMD. We found that the high expression of AF1q was an adverse prognostic factor in multiple myeloma.

Session topic: E-poster
Keyword(s): Multiple myeloma, Prognostic factor, Stem cell transplant
Abstract: E1282
Type: Eposter Presentation
Background
Multiple myeloma is an intractable hematological malignancy with various clinical manifestations, especially extramedullary disease (EMD). The prognosis of patients with EMD is extremely poor due to limited treatment options and aggressive nature of EMD, however the mechanism of the progression of EMD is not well known.AF1q is an oncogene which expressed in leukemia cells, located in 1q21. The gene is well known as one of the fusion partners of MLL, and as a poor prognostic factor in acute myeloid leukemia and myelodysplastic syndrome . Recently, we reported that high expression of AF1q results in an autonomous Wnt activity that promotes distant metastasis of breast cancer.
Aims
We hypothesized that high expression of AF1q is a poor prognostic factor of multiple myeloma and associated with EMD.To evaluate our hypothesis, we analyzed the expression of AF1q in patients with multiple myeloma and investigated the impact on the prognosis and the progression of EMD.
Methods
Newly diagnosed multiple myeloma patients in National Center for Global Health and Medicine hospital from January 2001 to March 2013 were studied. Patients were treated with vincristine-adriamycin-dexamethason or bortezomib-dexamethason induction therapy followed by autologous stem cell transplantation using high dose melphalan.The expression of AF1q was evaluated using the immunostaining of bone marrow clot samples at the diagnosis of multiple myeloma. The expression of AF1q was graded from “-“ to “+++”. EMD was diagnosed by pathological examination and/or CT/MRI/PET. The clinical response, progression free survival (PFS), and overall survival (OS) were analyzed using Kaplan Meier method with Log-rank test. Correlation between EMD and AF1q expression was tested by Chi-square test.
Results
Clinical data and bone marrow clot samples of 117 patients were analyzed. Mean age was 54.9 years old, and 51.9% was male. The grades of AF1q expression were 7 of “-”, 33 of “+”, 35 of “++”, and 42 of “+++”. We defined the cases with “-” and “+” as low expression, “++” and “+++” as high expression. Very good partial response or better was obtained after completion of autologous stem cell transplantation in 42.5% of patients with low AF1q expression and 37.7% of high expression. There was no statistically significant difference. Survival analysis using Log Rank method showed that high expression of AF1q was associated with significantly shorter progression free survival (3 year PFS was 56.2% in low AF1q expression vs. 30.5% in high AF1q expression, p=0.009), but not overall survival (3 year OS was 89.6% in low AF1q expression vs. 73.3% in high AF1q expression, ns. ).EMD was found in 25.0% of patients with low AF1q expression and 44.7% of high expression. Chi-square test showed that the incidence of EMD was significantly higher in patients with high AF1q expression than low expression (p=0.045).
Conclusion
To our knowledge, this is the first study that demonstrates a molecular marker associated with myeloma with EMD. We found that the high expression of AF1q was an adverse prognostic factor in multiple myeloma.

Session topic: E-poster
Keyword(s): Multiple myeloma, Prognostic factor, Stem cell transplant
Type: Eposter Presentation
Background
Multiple myeloma is an intractable hematological malignancy with various clinical manifestations, especially extramedullary disease (EMD). The prognosis of patients with EMD is extremely poor due to limited treatment options and aggressive nature of EMD, however the mechanism of the progression of EMD is not well known.AF1q is an oncogene which expressed in leukemia cells, located in 1q21. The gene is well known as one of the fusion partners of MLL, and as a poor prognostic factor in acute myeloid leukemia and myelodysplastic syndrome . Recently, we reported that high expression of AF1q results in an autonomous Wnt activity that promotes distant metastasis of breast cancer.
Aims
We hypothesized that high expression of AF1q is a poor prognostic factor of multiple myeloma and associated with EMD.To evaluate our hypothesis, we analyzed the expression of AF1q in patients with multiple myeloma and investigated the impact on the prognosis and the progression of EMD.
Methods
Newly diagnosed multiple myeloma patients in National Center for Global Health and Medicine hospital from January 2001 to March 2013 were studied. Patients were treated with vincristine-adriamycin-dexamethason or bortezomib-dexamethason induction therapy followed by autologous stem cell transplantation using high dose melphalan.The expression of AF1q was evaluated using the immunostaining of bone marrow clot samples at the diagnosis of multiple myeloma. The expression of AF1q was graded from “-“ to “+++”. EMD was diagnosed by pathological examination and/or CT/MRI/PET. The clinical response, progression free survival (PFS), and overall survival (OS) were analyzed using Kaplan Meier method with Log-rank test. Correlation between EMD and AF1q expression was tested by Chi-square test.
Results
Clinical data and bone marrow clot samples of 117 patients were analyzed. Mean age was 54.9 years old, and 51.9% was male. The grades of AF1q expression were 7 of “-”, 33 of “+”, 35 of “++”, and 42 of “+++”. We defined the cases with “-” and “+” as low expression, “++” and “+++” as high expression. Very good partial response or better was obtained after completion of autologous stem cell transplantation in 42.5% of patients with low AF1q expression and 37.7% of high expression. There was no statistically significant difference. Survival analysis using Log Rank method showed that high expression of AF1q was associated with significantly shorter progression free survival (3 year PFS was 56.2% in low AF1q expression vs. 30.5% in high AF1q expression, p=0.009), but not overall survival (3 year OS was 89.6% in low AF1q expression vs. 73.3% in high AF1q expression, ns. ).EMD was found in 25.0% of patients with low AF1q expression and 44.7% of high expression. Chi-square test showed that the incidence of EMD was significantly higher in patients with high AF1q expression than low expression (p=0.045).
Conclusion
To our knowledge, this is the first study that demonstrates a molecular marker associated with myeloma with EMD. We found that the high expression of AF1q was an adverse prognostic factor in multiple myeloma.

Session topic: E-poster
Keyword(s): Multiple myeloma, Prognostic factor, Stem cell transplant
{{ help_message }}
{{filter}}


