NEOD001 DEMONSTRATES CARDIAC AND RENAL BIOMARKER RESPONSES IN A PHASE 1/2 STUDY IN PATIENTS WITH AL AMYLOIDOSIS AND PERSISTENT ORGAN DYSFUNCTION
(Abstract release date: 05/21/15)
EHA Library. Liedtke M. 06/12/15; 103131; S104
Disclosure(s): Stanford University School of Medicine
Michaela Liedtke
Contributions
Contributions
Abstract
Abstract: S104
Type: Oral Presentation
Presentation during EHA20: From 12.06.2015 12:15 to 12.06.2015 12:30
Location: Room A2+3
Background
Light chain (AL) amyloidosis results from an accumulation of misfolded proteins that cause the dysfunction of vital organs (eg, heart and kidneys). Current therapeutic approaches target the plasma cells that produce the pathogenic light chain proteins and are typically associated with significant adverse effects. There is a substantial need for a safe and effective therapy that specifically targets the misfolded proteins responsible for the underlying organ dysfunction. NEOD001, a monoclonal antibody that targets these misfolded proteins, is hypothesized to neutralize circulating soluble protein aggregates and to clear insoluble aggregates from organs. We report data from a phase 1/2 dose-escalation/expansion study of NEOD001 in patients with AL amyloidosis and persistent organ dysfunction (NCT01707264).
Aims
The primary aims of this study were to determine the maximum tolerated dose/recommended phase 2 dose (RP2D) and the safety and tolerability of single-agent NEOD001 when administered to patients with AL amyloidosis. Secondary and exploratory objectives included pharmacokinetics (PK), immunogenicity, and hematologic and best organ responses based on consensus criteria.
Methods
Patients who completed ≥1 previous anti–plasma cell systemic therapy, had partial response or better, did not require additional chemotherapy, and had persistent organ dysfunction received NEOD001 intravenously every 28 days (q28d). Dose levels of 0.5, 1, 2, 4, 8, 16, and 24 mg/kg were evaluated in a 3+3 study design. Informed consent was obtained from all patients.
Results
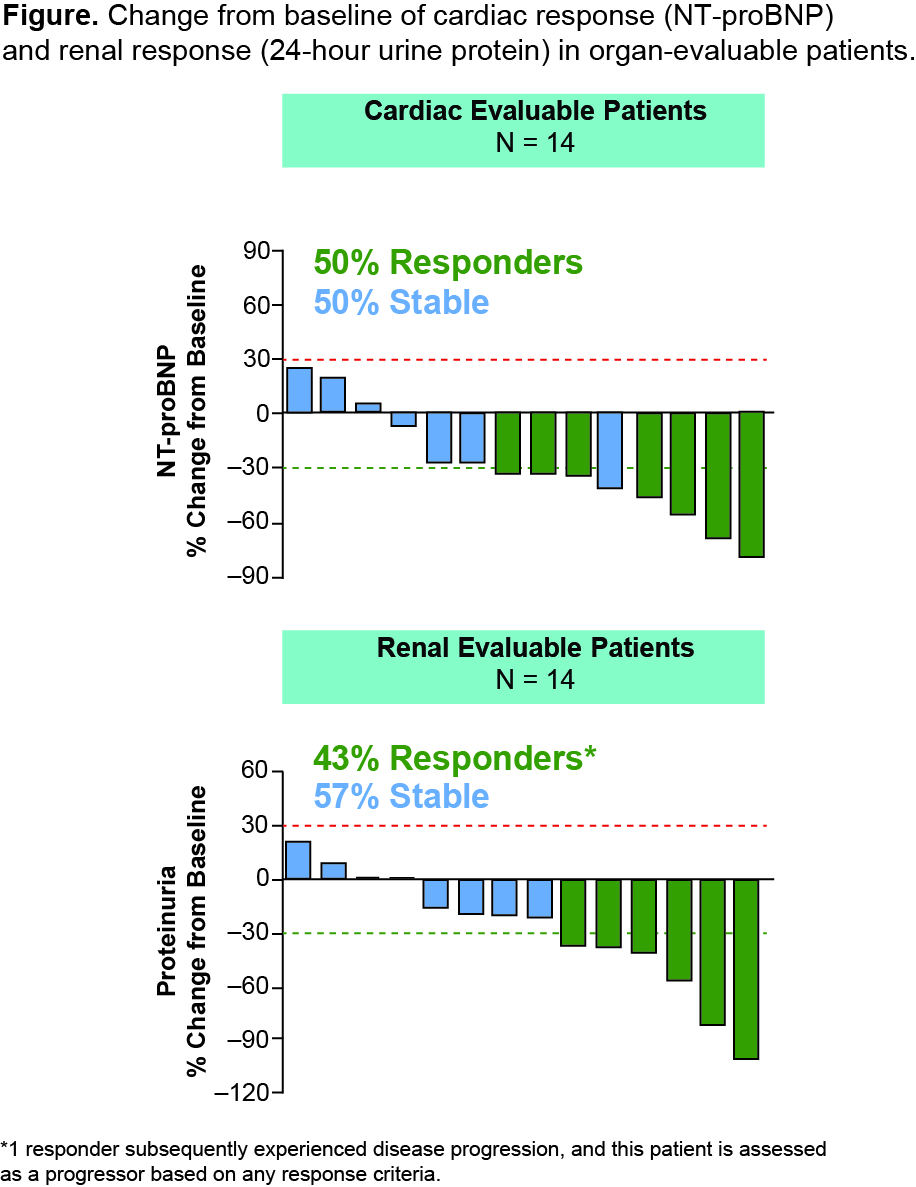
As of September 30, 2014, 27 patients in 7 cohorts received 209 infusions. Mean treatment duration was 8 months. No deaths, drug-related serious adverse events (AEs), discontinuations due to drug-related AEs, dose-limiting toxicities, or antidrug antibodies were reported. The most frequently reported AEs were fatigue, cough, and dyspnea. 24 mg/kg was selected as the RP2D. PK data support intravenous dosing q28d. Of the 14 patients evaluable for cardiac biomarker assessment, 50% met criteria for cardiac response (NT-proBNP: 30% reduction), and 50% achieved disease stabilization (Figure). Of the 14 renal evaluable patients, 43% met criteria for renal response (24-hour urine protein: 30% reduction), and 57% achieved disease stabilization (Figure).
Summary
Monthly infusions of NEOD001 were safe and well tolerated. 24 mg/kg was the RP2D. The cardiac response rate was 50%, and the renal response rate was 43%. These organ response rates compare favorably to those reported with traditional chemotherapy. The phase 2 expansion phase is ongoing. A phase 3 study has been initiated. Antibody therapy may represent a new therapeutic platform for the management of AL amyloidosis.
Keyword(s): Amyloidosis, Monoclonal antibody
Session topic: Multiple myeloma: Clinical studies 1
Type: Oral Presentation
Presentation during EHA20: From 12.06.2015 12:15 to 12.06.2015 12:30
Location: Room A2+3
Background
Light chain (AL) amyloidosis results from an accumulation of misfolded proteins that cause the dysfunction of vital organs (eg, heart and kidneys). Current therapeutic approaches target the plasma cells that produce the pathogenic light chain proteins and are typically associated with significant adverse effects. There is a substantial need for a safe and effective therapy that specifically targets the misfolded proteins responsible for the underlying organ dysfunction. NEOD001, a monoclonal antibody that targets these misfolded proteins, is hypothesized to neutralize circulating soluble protein aggregates and to clear insoluble aggregates from organs. We report data from a phase 1/2 dose-escalation/expansion study of NEOD001 in patients with AL amyloidosis and persistent organ dysfunction (NCT01707264).
Aims
The primary aims of this study were to determine the maximum tolerated dose/recommended phase 2 dose (RP2D) and the safety and tolerability of single-agent NEOD001 when administered to patients with AL amyloidosis. Secondary and exploratory objectives included pharmacokinetics (PK), immunogenicity, and hematologic and best organ responses based on consensus criteria.
Methods
Patients who completed ≥1 previous anti–plasma cell systemic therapy, had partial response or better, did not require additional chemotherapy, and had persistent organ dysfunction received NEOD001 intravenously every 28 days (q28d). Dose levels of 0.5, 1, 2, 4, 8, 16, and 24 mg/kg were evaluated in a 3+3 study design. Informed consent was obtained from all patients.
Results
As of September 30, 2014, 27 patients in 7 cohorts received 209 infusions. Mean treatment duration was 8 months. No deaths, drug-related serious adverse events (AEs), discontinuations due to drug-related AEs, dose-limiting toxicities, or antidrug antibodies were reported. The most frequently reported AEs were fatigue, cough, and dyspnea. 24 mg/kg was selected as the RP2D. PK data support intravenous dosing q28d. Of the 14 patients evaluable for cardiac biomarker assessment, 50% met criteria for cardiac response (NT-proBNP: 30% reduction), and 50% achieved disease stabilization (Figure). Of the 14 renal evaluable patients, 43% met criteria for renal response (24-hour urine protein: 30% reduction), and 57% achieved disease stabilization (Figure).
Summary
Monthly infusions of NEOD001 were safe and well tolerated. 24 mg/kg was the RP2D. The cardiac response rate was 50%, and the renal response rate was 43%. These organ response rates compare favorably to those reported with traditional chemotherapy. The phase 2 expansion phase is ongoing. A phase 3 study has been initiated. Antibody therapy may represent a new therapeutic platform for the management of AL amyloidosis.
Keyword(s): Amyloidosis, Monoclonal antibody

Session topic: Multiple myeloma: Clinical studies 1
Abstract: S104
Type: Oral Presentation
Presentation during EHA20: From 12.06.2015 12:15 to 12.06.2015 12:30
Location: Room A2+3
Background
Light chain (AL) amyloidosis results from an accumulation of misfolded proteins that cause the dysfunction of vital organs (eg, heart and kidneys). Current therapeutic approaches target the plasma cells that produce the pathogenic light chain proteins and are typically associated with significant adverse effects. There is a substantial need for a safe and effective therapy that specifically targets the misfolded proteins responsible for the underlying organ dysfunction. NEOD001, a monoclonal antibody that targets these misfolded proteins, is hypothesized to neutralize circulating soluble protein aggregates and to clear insoluble aggregates from organs. We report data from a phase 1/2 dose-escalation/expansion study of NEOD001 in patients with AL amyloidosis and persistent organ dysfunction (NCT01707264).
Aims
The primary aims of this study were to determine the maximum tolerated dose/recommended phase 2 dose (RP2D) and the safety and tolerability of single-agent NEOD001 when administered to patients with AL amyloidosis. Secondary and exploratory objectives included pharmacokinetics (PK), immunogenicity, and hematologic and best organ responses based on consensus criteria.
Methods
Patients who completed ≥1 previous anti–plasma cell systemic therapy, had partial response or better, did not require additional chemotherapy, and had persistent organ dysfunction received NEOD001 intravenously every 28 days (q28d). Dose levels of 0.5, 1, 2, 4, 8, 16, and 24 mg/kg were evaluated in a 3+3 study design. Informed consent was obtained from all patients.
Results
As of September 30, 2014, 27 patients in 7 cohorts received 209 infusions. Mean treatment duration was 8 months. No deaths, drug-related serious adverse events (AEs), discontinuations due to drug-related AEs, dose-limiting toxicities, or antidrug antibodies were reported. The most frequently reported AEs were fatigue, cough, and dyspnea. 24 mg/kg was selected as the RP2D. PK data support intravenous dosing q28d. Of the 14 patients evaluable for cardiac biomarker assessment, 50% met criteria for cardiac response (NT-proBNP: 30% reduction), and 50% achieved disease stabilization (Figure). Of the 14 renal evaluable patients, 43% met criteria for renal response (24-hour urine protein: 30% reduction), and 57% achieved disease stabilization (Figure).
Summary
Monthly infusions of NEOD001 were safe and well tolerated. 24 mg/kg was the RP2D. The cardiac response rate was 50%, and the renal response rate was 43%. These organ response rates compare favorably to those reported with traditional chemotherapy. The phase 2 expansion phase is ongoing. A phase 3 study has been initiated. Antibody therapy may represent a new therapeutic platform for the management of AL amyloidosis.
Keyword(s): Amyloidosis, Monoclonal antibody

Session topic: Multiple myeloma: Clinical studies 1
Type: Oral Presentation
Presentation during EHA20: From 12.06.2015 12:15 to 12.06.2015 12:30
Location: Room A2+3
Background
Light chain (AL) amyloidosis results from an accumulation of misfolded proteins that cause the dysfunction of vital organs (eg, heart and kidneys). Current therapeutic approaches target the plasma cells that produce the pathogenic light chain proteins and are typically associated with significant adverse effects. There is a substantial need for a safe and effective therapy that specifically targets the misfolded proteins responsible for the underlying organ dysfunction. NEOD001, a monoclonal antibody that targets these misfolded proteins, is hypothesized to neutralize circulating soluble protein aggregates and to clear insoluble aggregates from organs. We report data from a phase 1/2 dose-escalation/expansion study of NEOD001 in patients with AL amyloidosis and persistent organ dysfunction (NCT01707264).
Aims
The primary aims of this study were to determine the maximum tolerated dose/recommended phase 2 dose (RP2D) and the safety and tolerability of single-agent NEOD001 when administered to patients with AL amyloidosis. Secondary and exploratory objectives included pharmacokinetics (PK), immunogenicity, and hematologic and best organ responses based on consensus criteria.
Methods
Patients who completed ≥1 previous anti–plasma cell systemic therapy, had partial response or better, did not require additional chemotherapy, and had persistent organ dysfunction received NEOD001 intravenously every 28 days (q28d). Dose levels of 0.5, 1, 2, 4, 8, 16, and 24 mg/kg were evaluated in a 3+3 study design. Informed consent was obtained from all patients.
Results
As of September 30, 2014, 27 patients in 7 cohorts received 209 infusions. Mean treatment duration was 8 months. No deaths, drug-related serious adverse events (AEs), discontinuations due to drug-related AEs, dose-limiting toxicities, or antidrug antibodies were reported. The most frequently reported AEs were fatigue, cough, and dyspnea. 24 mg/kg was selected as the RP2D. PK data support intravenous dosing q28d. Of the 14 patients evaluable for cardiac biomarker assessment, 50% met criteria for cardiac response (NT-proBNP: 30% reduction), and 50% achieved disease stabilization (Figure). Of the 14 renal evaluable patients, 43% met criteria for renal response (24-hour urine protein: 30% reduction), and 57% achieved disease stabilization (Figure).
Summary
Monthly infusions of NEOD001 were safe and well tolerated. 24 mg/kg was the RP2D. The cardiac response rate was 50%, and the renal response rate was 43%. These organ response rates compare favorably to those reported with traditional chemotherapy. The phase 2 expansion phase is ongoing. A phase 3 study has been initiated. Antibody therapy may represent a new therapeutic platform for the management of AL amyloidosis.
Keyword(s): Amyloidosis, Monoclonal antibody

Session topic: Multiple myeloma: Clinical studies 1
{{ help_message }}
{{filter}}


